Brain monitoring built for the full surgical journey.
Drug-aware brain monitoring across pre-op baseline, intra-op depth, and post-op recovery. One system for the full surgical journey.
Drug-aware brain monitoring across pre-op baseline, intra-op depth, and post-op recovery. One system for the full surgical journey.
1 in 5 American adults has preclinical Alzheimer’s. Most don’t know it — and anesthesia may be the trigger.
of older surgical patients develop delirium or cognitive decline after anesthesia.
added each year to the U.S. healthcare system from perioperative brain complications.
live with preclinical Alzheimer’s — undetected before they go under anesthesia.
Sources: Cognitive decline · Cost effectiveness for delirium · Brain-function-monitoring · Alzheimer’s disease · MIT Brain Health Study
ML-driven cognitive screening establishes a brain-health baseline and flags preclinical vulnerability before the first drug is given.
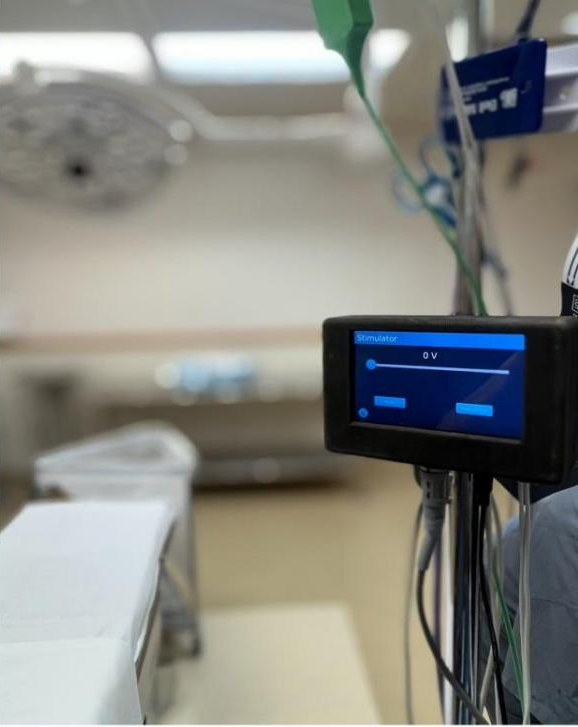
Real-time multi-region EEG and AEP, interpreted by proprietary ML, delivering depth-of-sedation monitoring that stays reliable across all major anesthesia agents and mixed-drug sedation.
Continuous post-anesthesia monitoring detects emergent delirium and forecasts recovery trajectory before discharge.
The KBO platform combines a wearable EEG headband and in-ear auditory probe with a clinical monitor that displays real-time brain-state metrics. Currently undergoing IRB-approved study at UC Davis and McLeod Health.


11+ years in medical device sales and commercialization at Medtronic, Philips, and Stryker. Hospital relationships across 8 states. Prior startup operator.

Professor and Chair of Anesthesiology, UC Davis Health. National leader in cardiac anesthesia and perioperative brain health. 20+ years clinical leadership.

Brings expertise in anesthesiology and cognitive neuroscience from his academic work at UC Davis. Collaboration is advisory.

Chief of Anesthesia at McLeod Health. Leads multi-site data collection and clinical protocol design. Former ICU nurse at Johns Hopkins.




